Crystal System: There are
230-crystal forms
32-point groups
7- Crystal systems
14- Bravais Lattice
There are seven types of
unit cell formed. These are Cubic, Tetragonal, Orthorhombic, Monoclinic,
Hexagonal, Rhombohedral or Trigonal and Triclinic. The 7 crystal systems can be remembered
as C T
O M T
R H
Crystal System
|
Relation between a, b, c,
|
Relation between α, β, ɣ
|
No. of Planes and Axes
|
Cubic
 |
a = b = c
|
α = β = ɣ = 90⁰
|
9 planes + 4
axes
|
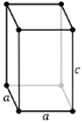
Tetragonal
 |
a = b ≠ c
|
α = β = ɣ = 90⁰
|
5 planes + 5 axes
|
Orthorhombic
 |
a ≠ b ≠ c
|
α = β = ɣ = 90⁰
|
3 planes + 3
axes
|
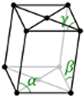
Monoclinic
 |
a ≠ b ≠ c
|
α = β = 90⁰ ≠ ɣ
|
1 plane + 1
axis
|
Triclinic
 |
a ≠ b ≠ c
|
α ≠ β ≠ ɣ ≠ 90⁰
|
0
|
Rhombohedral
 |
a = b = c
|
α = β = ɣ ≠ 90⁰
|
7 planes + 7
axes
|
Hexagonal
 |
a = b ≠ c
|
α = β = 90⁰, ɣ
= 120⁰
|
7 planes + 7 axes
|
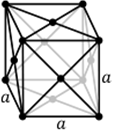
Bravais lattices: The Bravais lattice are the distinct lattice types which when
repeated can fill the whole space. In three dimensions there are 14 Bravais lattices
Primitive
|
Side Centered
|
Body Centered
|
Face Centered
|
 |
 |
 |
|
Cubic
|
|||
a ≠ c
 |
a ≠ c
 |
||
Tetragonal
|
|||
a ≠ b ≠ c
 |
a ≠ b ≠ c
 |
a ≠ b ≠ c
 |
a ≠ b ≠ c
 |
Orthorhombic
|
|||
α ≠ 90⁰, β, γ =
90⁰
 |
α ≠ 90⁰, β, γ =
90⁰
 |
||
Monoclinic
|
|||
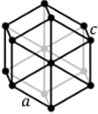 Hexagonal |
α = β = γ ≠ 90⁰

Trigonal
|
α, β, γ ≠ 90⁰

Triclinic
|
|
Crystal
|
Description
|
Co. Number
|
Rock Salt
(NaCl)
|
Cl⁻
ions are in CCP and Na⁺ ions occupy
octahedral voids
|
Cl⁻
→ 6
Na⁺ → 6
|
Zincbled (ZnS)
|
S²⁻
ions are in CCP Zn²⁺ ions occupied
tetrahedral voids.
|
Zn²⁺ → 4
S²⁻ → 4
|
CsCl
|
Cl⁻
ions are in HCP Cs⁺ ions in the body of the cube.
|
Cl⁻ → 8
Cs⁺ → 8
|
Wurtzrite (ZnS)
|
S²⁻
ions are in HCP Zn²⁺ ions occupies
tetrahedral void.
|
Zn²⁺ → 4
S²⁻ → 4
|
Florite (CaF₂)
(AB₂)
|
Ca²⁺
ions are in CCP F⁻ ions occupies all the tetrahedral voids
|
Ca²⁺ → 8
F⁻ → 4
|
Anti Florit
(A₂B)
|
A⁺
ions occupies all the tetrahedral voids, B²⁻
ions are in CCP
|
A⁺ → 4
B²⁻ → 8
|
Structure of CsCl:

1. Total no: of ions = 9 (1Cs⁻ + 8 Cl⁻
[Or] 1 Cl⁻ + 8 Cs⁺)
2. No: of Cs⁺ per unit cell = 0/8 +
0/4 + 0/2 + 1/1 = 1
3. No: of Cl⁻ per unit cell = 8/8 +
0/4 + 0/2 + 0/1 = 1
4. No: of CsCl formula units per unit
cell = 1.
5. Co-ordination number
Each ion is surrounded by eight ions of its
counterpart.
So, coordination number for
Cs⁺ → 8
Cl⁻ → 8
6. Body diagonal = √3a = 2 (r Cs⁺
+ r Cl⁻)
Structure of NaCl:

a. Total no: of ions = 27 (13 Na⁺ + 14
Cl⁻ [Or] 14 Na⁺ + 13 Cl⁻)
b. No: of Na⁺ ions per unit cell = 8/8
+ 12/4 = 4 {1 is at body centered 12 are at edges}
c. No: of Cl⁻ ions per unit cell = 6/2
+ 8/8 = 3 {8 at corners and 6 at face center}
d. No: of formula units per unit cell =
4
e. Co-ordination no: of Na⁺ = 6
f. Cl⁻ = 6
g. Edge length = a = 2 (rNa⁺
+ rCl⁻)
No comments:
Post a Comment